The oxygen has a formal charge of 6 - 4 - 2 = 0 (same ordering of terms). The central carbon has a formal charge of 4 (valence electrons) - 0 (lone pairs) - 4 (bonds) = 0. Use formal charges to identify the most reasonable Lewis structure for a given molecule. All atoms in the molecule have zero formal charge, the "happiest" situation for any molecule. Compute formal charges for atoms in any Lewis structure. Each hydrogen atom has one electron as a neutral atom, no lone pairs and shares one bond, for a formal charge of zero. It has no lone pairs, and it shares four bonds, so the formal charge is zero. The carbon in CH 4 has four electrons as a neutral atom. Subtract the number of bonds shared by the atom.Subtract the number of non-bonding electrons (usually in lone pairs). Calculating Formal Charge from Lewis Structures Assign formal charges to each atom in the interhalogen ion ICl4. 
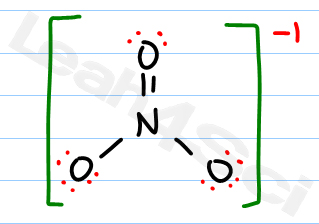
Count the number of valence electrons of the neutral atom.The sum of the formal charges, with a couple of extra rules, will help us to decide which of multiple-possible valid Lewis structures is likely to be the correct one. Formal charge is just a way of bookkeeping that helps us to decide which of multiple Lewis structures is the likely true bonding arrangement of a covalent molecule. In those cases we resort to calculating what's called the formal charge of each atom. Resonance form 1 is the most stable and thus the most preferred Lewis representation of NCO because, in this, the -1 formal charge is located on the most electronegative O-atom. Sometimes it's difficult to tell which of two possible Lewis structures of a compound represents the actual bonding of the molecule. A -1 overall formal charge is present on each of the above resonance structures.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
